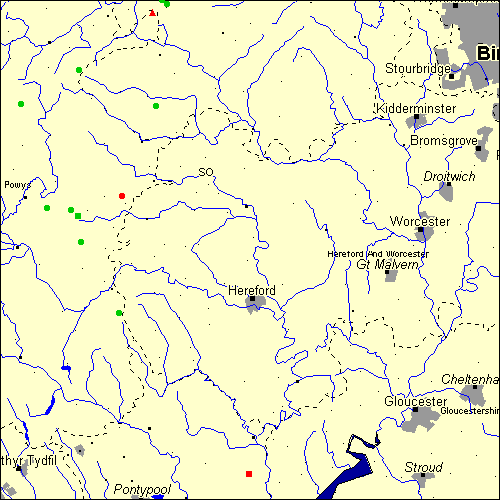
Stone Circles and Rows on Map Sheet SO
SJ |
||
SN |
| SP |
ST |
Move the mouse over a point on the map to see the Stone Circle's name and map reference in the status bar.
SJ |
||
SN |

| SP |
ST |
Move the mouse over a point on the map to see the Stone Circle's name and map reference in the status bar.
The Map sheet SO covers a 100 km2 area which includes the following stone circles and rows:
Banc Du Stone Circle at map reference SO042792. Four Stones Stone Circle at map reference SO245607. www Gelli Hill Stone Circle at map reference SO095583. Harolds Stones Stone Row(s) at map reference SO499051. www Hoarstones Stone Circle at map reference SO324999. Kerry Hill Stone Circle at map reference SO158860. Pedwar Maen Stone Row(s) at map reference SO156568. Pen Y Beacon Stone Circle at map reference SO239373. Pen-Y-Wern Hill Stone Circle at map reference SO313788. Rhos-Maen Stone Circle at map reference SO143579. Shelve Stone Circle at map reference SO335992. Six Stones Stone Circle at map reference SO163516. Whetstones group at map reference SO305976. www The name and map reference of the megaliths appear in the status bar when the mouse pointer is placed over a point on the map above. In each map reference, the letters SO refer to the 100 km2 sheet shown above. The next three numbers are the distance across from the left edge (0 to 999)
and the final three numbers are the distance up from the bottom edge, (0 to 999). A point on the map in red has further details on another web site, a point in green does not Circles on the map represent stone circles, squares represent single or multiple stone rows or avenues, triangles represent group sites
with two or more megalithic sites close together.
 |
The list of stone circles and rows included in this resource is now available for browsing alphabetically. |
 |
Any stone circle or row indicated on the above map has further details given on a separate page here at the Megalith Map. Circles and rows indicated by a www tag, have links to websites elsewhere on the internet containing more text and/or images. Tell us about a stone circle or row not included on the above map by dropping us a line, and helping us to make this resource better for all who use it.
Go back to the map of the British Isles.
This information is brought to you by Andy and Alastair's Cheeseless ProductionsTM. This page published on 03/10/98 22:08:18.